 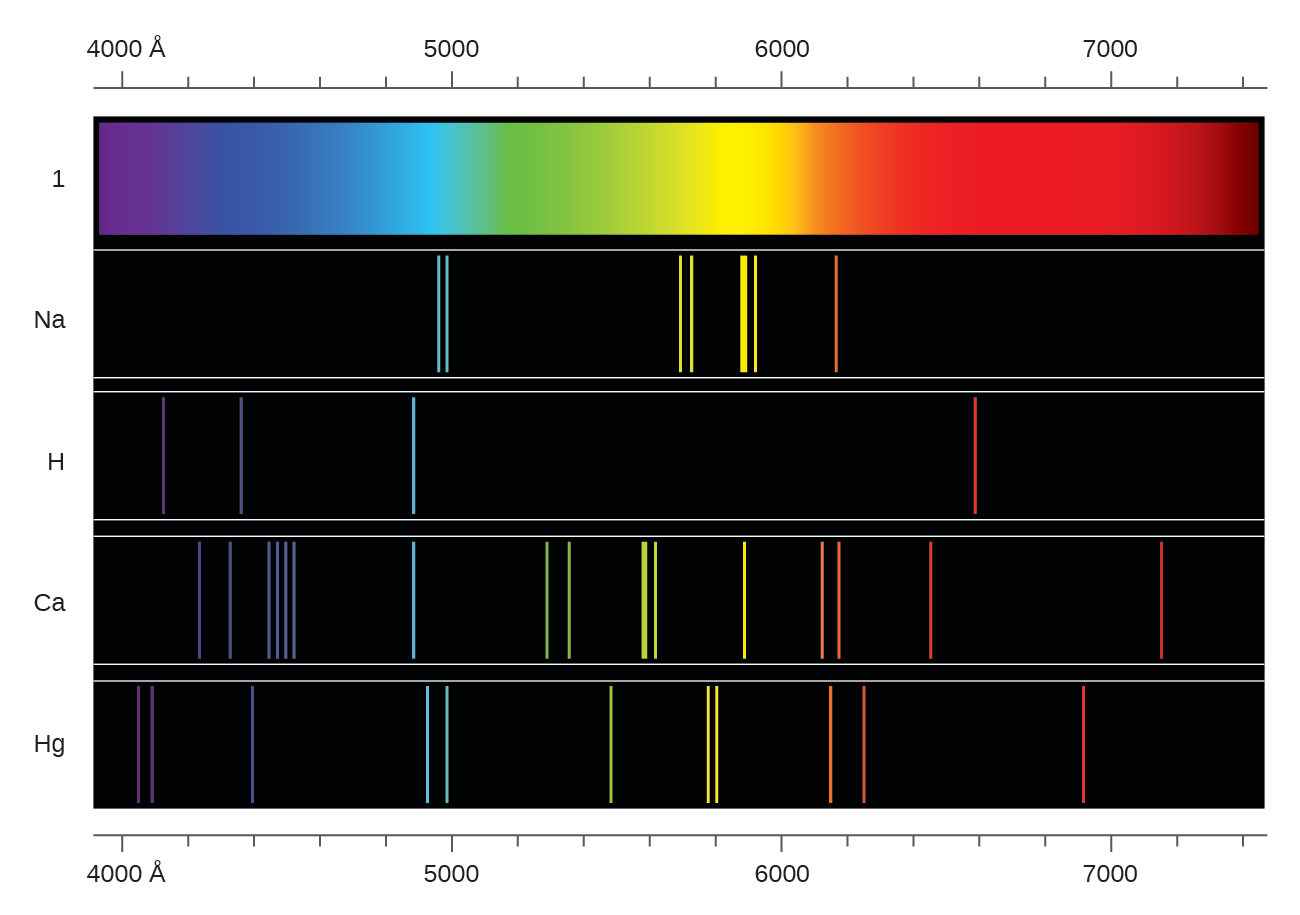
The helium emission spectrum is produced by the emission of light by helium atoms in excited states. The hydrogen emission spectrum is produced here by the emission of light by hydrogen atoms in excited states. The emission spectrum is characterized by a sequence of lines on a black backdrop. It is mostly due to the fact that hydrogen contains one electron per atom, but helium has two electrons per atom.įurthermore, there is a significant difference between hydrogen and helium emission spectra in that electron-electron repulsions have no effect on hydrogen emission spectra due to the presence of a single electron in the hydrogen atom, whereas electron-electron repulsions affect helium emission spectra due to the presence of two electrons. The main distinction between hydrogen and helium emission spectra is that the helium emission spectrum contains more lines than the hydrogen emission spectrum. The helium emission spectrum, on the other hand, is a spectrum formed by the emission of light by helium atoms in excited states. The hydrogen emission spectrum is a spectrum formed by the emission of light by excited hydrogen atoms. The nonclassical notion that electrons move in particular shells, or orbits, around the nucleus underpins Bohr's hydrogen model. 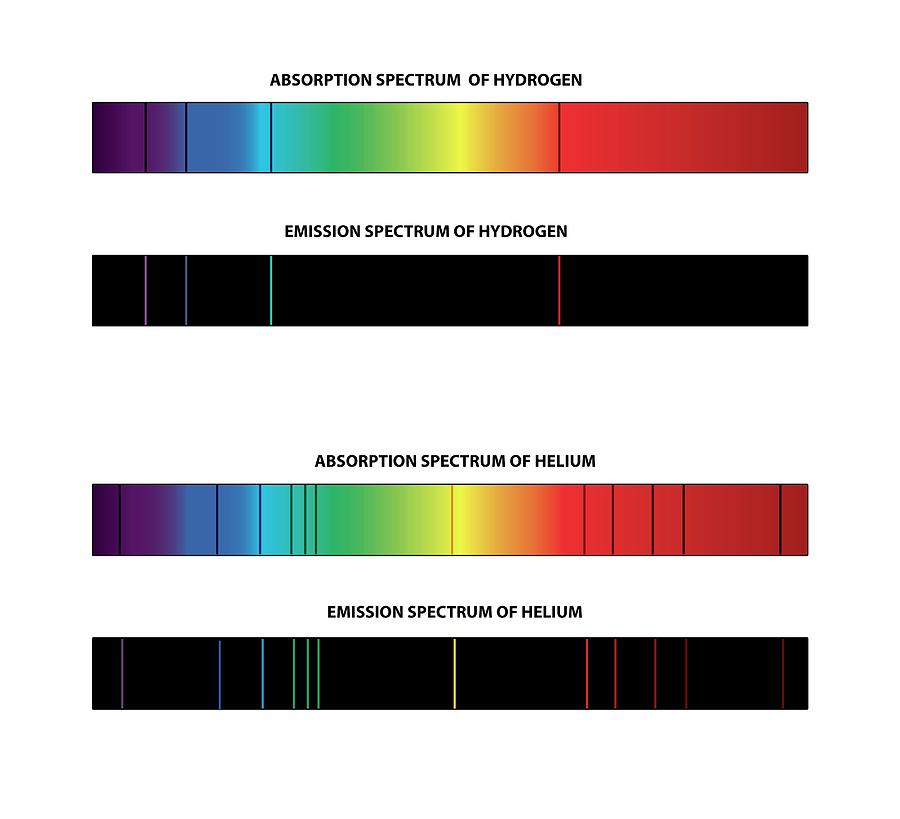
Molecular spectra are detected when a molecule absorbs or emits electromagnetic radiation, causing a rise or reduction in energy. Hint: We know that in order to answer this question, we must first understand the Bohr model for hydrogen and hydrogen-like substances and also the concept of spectrum. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
